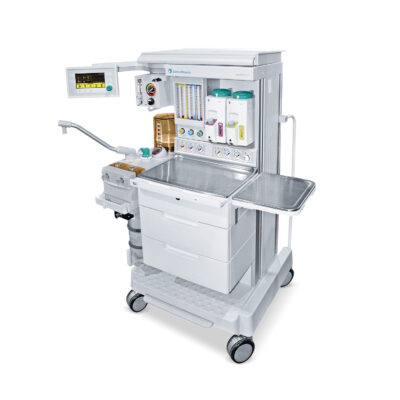
GE Aespire S5 Anesthesia Machine with 7900 Smartvent
The Aespire 7900 combines the exceptional performance of the 7900 SmartVent with advanced patient monitoring technology into an affordable anesthesia delivery solution that’s well-suited for the ambulatory surgery center.
Clinician-inspired solutions help improve patient care
Clinical inspired Ventilation
You asked for sophisticated ventilation capabilities that help you meet the needs of a broad patient range. The Aespire 7900 satisfies your request with the 7900 SmartVent ventilator. The SmartVent uses a similar gas delivery system that is found in most critical care ventilators, yet has been adapted specifically for anesthesia applications and is easily controlled via our intuitive user interface.
Ideal for spontaneously breathing patients
SIMV and PSVPro modes include an adjustable flow trigger, trigger window, electronic PEEP and an apnea backup mode to simplify the work of caring for your spontaneously breathing patients. Patients with laryngeal mask airways (LMAs) and those that cannot tolerate certain anesthetic agents can benefit from the use of SIMV and PSVPro modes from the use of SIMV and PSVPro modes.
A wide range of ventilation odes expands the clinical capabilities of the Aespire 7900 SmartVent to help meet your patient’s needs:
- Volume control
- Pressure Control
- Synchronized Intermittent Mandatory Ventilation (SIMV) with pressure Support
- PSVPro(Pressure Support with Apnea backup mode)
- The pressure Control Mode lets you chose a target pressure and deliver the maximum tidal volume for the desired time and pressure selected.
Physical Weight & Dimensions
| Physical Weight & Dimensions |
|
|---|---|
| Top Shelf |
|
| Work Surface |
|
| DIN Rail | Side of machine: 34.5 cm/13.6 in |
| Drawers (internal dimensions) |
|
| Absorber Bag Arm (optional) |
|
Electrical Specifications
| Current Leakage |
|
|---|---|
| Inlet/outlet modules (120 V) |
|
Pneumatic Specifications
| Auxiliary Common Gas Outlet | Connector: ISO 22 mm OD and 15 mm ID |
|---|---|
| Gas Supply |
|
Environmental Specifications
| System Operation |
|
|---|---|
| System Storage |
|
| Electromagnetic Compatibility |
|
For new products, please refer to the manufacturer’s website for details.
This product has been refurbished and tested to the highest quality standards by The PCB Medical. This Limited Warranty offered by PCB Medical covers defects in material or workmanship. This warranty extends to the original purchaser only and is nontransferable. The warranty begins the day the equipment is received. Please inspect your item(s) upon delivery. Any unauthorized service providers working on our equipment within the warranty period will void the warranty!
Our warranty periods are very based on product, a standard 30 day parts warranty is included with all refurbished parts unless stated otherwise on the quote/Invoice. Warranties begin from the documented date of receipt.
Our warranties do not cover any problem that is caused by: A. Conditions, malfunctions or damage not resulting from defects in material or workmanship. B. Conditions, malfunctions or damage resulting from (1) normal wear and tear, improper installation, improper maintenance, misuse, abuse, negligence, accident or alteration.
Our limited warranties are void if a product is returned with removed, damaged or tampered labels or any alterations (including removal of any component or external cover).
PCB Medical will not provide any warranty coverage unless claims are made in compliance with all terms of the controlling warranty statement included with your product and you follow proper return procedure. To request warranty service, you will need to provide: 1. The invoice or other evidence of the date and place of purchase. 2. A description of the problem. 3. Contact our Customer Service Department to receive a Return Authorization Number (RMA#)
Damaged freight needs to be reported within 24 hours to file a claim. No merchandise may be returned without prior written authorization by PCB Medical.
No NEW merchandise may be returned unless authorization from manufacturer which could result in a restocking fee or no return at all, and customer is responsible for shipping expenses.
Return period for products is thirty (30) days from delivery or otherwise agreed upon.
The PCB Medical will test returned products returned and can impose a customer fee if the customer misrepresents the condition of the product.
Credit & Return Shipping Guidelines
Order cancellations (after 24 hours) and returns are charged a 45% restocking fee. Damaged goods will receive credit dependent on the condition of the product, and customer is responsible for shipping expenses.
No credit will be issued if the item is non-returnable, such as a new product, custom or special order, or in the case that the item is damaged due to improper handling/packaging by the customer.
Shipping Charges: Customers are responsible for making sure items can be delivered into or outside of their facility without issue. For example, can a 53’ foot truck access your building? Failure to accommodate delivery can result in items not being delivered, additional fees, or return of equipment.
Basic Shipping Charge is for dock to dock shipping and/or liftgate service shipping. The items shipping dock to dock and/or liftgate are skidded/palletized items. It doesn’t not include inside delivery.
Residential shipping charge is for customer’s needing delivery in a residential area.
Inside delivery is for customer’s needing their purchase delivered inside the building. The item(s) will be delivered into the lobby only. They are not delivered into rooms/suites, elevators, or stairs.
Customers are responsible for disposing of packing materials.
If you are needing white glove delivery, please contact us directly at info@pcbhme.com